Description
The Alzheimer amyloid precursor protein (APP) is a transmembrane protein whose abnormal processing is associated with the pathogenesis of Alzheimer’s disease. APP695 lacking the protease inhibitor domain is the predominant form in neuronal tissues. APP695 is cleaved by caspases into the 664-residue amino (N)-terminal fragment that lacks the carboxyl C-terminal 31-residues (APPΔC31) and the 31-residues C-terminal fragment (APP-C31). Both fragments might be potent inducers of neuronal apoptosis. An antibody (named ACT1) against the N-terminus of caspase 3-generated APP C-terminal 31 aa of human APP695 (APP-C31) was raised in rabbit.
Applications
Western blot (dilution: 1/3,000-1/1,000)
Immunocytochemistry (dilution: 1/1,000-1/500)
ELISA
Other applications have not been tested.
Specification
Immunogen: Synthetic peptide corresponding to the C-terminus of the caspase 3-cleaved human APP (aa 658-664 of human APP695)
Specificity: Specific to the C-terminal end of APP ΔC31
Form: Antiserum with 0.05% sodium azide
Storage: Shipped at 4°C and stored at -20°C
Data Link:
UniProtKB/Swiss-Prot P05067 (A4_HUMAN)
References: This antibody was used in ref. 3.
Kang HG et al (1987) “The precursor of Alzheimer’s disease amyloid A4 protein resembles a cell-surface receptor.” Nature 325: 33-736 PMID: 2881207
Selkoe DJ (1994) “Normal and abnormal biology of the beta-amyloid precursor protein.” Annu Rev Neurosci 17: 489-517 PMID: 8210185
Nishimura I et al (2002) “Cell death induced by a caspase-cleaved transmembrane fragment of the Alzheimer amyloid precursor protein.” Cell Death Differ 9: 199-208 PMID: 11840170
Nishimura I et al (2003) “Upregulation and antiapoptotic role of endogenous Alzheimer amyloid precursor protein in dorsal root ganglion neurons.” Exp Cell Res 286: 241-251 PMID: 12749853
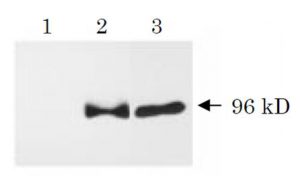
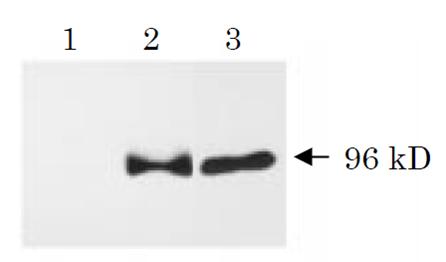
Fig. 1 Western blot analysis of APPΔC31 (ref. 3). Human NT2 neurons (neurally differentiated human NT2 embryonic carcinoma cells) were infected with adenovirus vector expressing β-galactosidase (lane 1), wild-type APP (lane 2) or APPΔC31 (lane3). Cell lysates were prepared 48 h after infection, and proteins were analyzed by Western blot using this antibody (SAC). Neurons overexpressing wild-type APP contained a 96 kD SAC-immunoreactive fragment which was also detected in APPΔC31-overexpressing neurons.
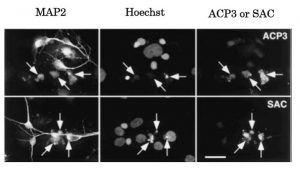
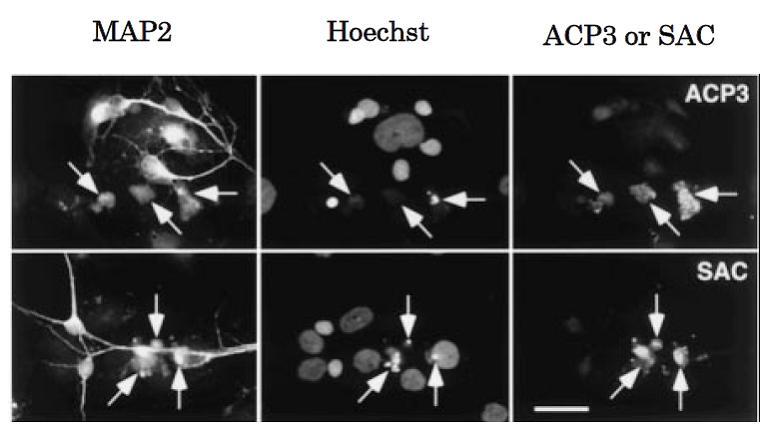
Fig. 2 Immunocytochemical analysis of APPΔC31: Caspase-3 activation and generation of the caspase-cleaved fragment APPΔC31 within neurons induced by serum deprivation (ref. 3). Neurally differentiated NT2 cells were cultured for 96 h in the absence of fetal calf serum. Cells were triply labeled for the neuronal marker microtubule-associated protein 2 (MAP2), chromosomal DNA (Hoechst), and activated caspase-3 (ACP3; upper panel) or APPΔC31 (SAC; lower panel). MAP2-immunopositive neurons with apoptotic nuclei (arrows) are intensively immunostained with ACP3 and SAC.