Description
ATF6 (activating transcription factor 6) is an endoplasmic reticulum (ER) membrane-bound transcription factor activated in response to ER stress. When unfolded proteins accumulate in the ER, ATF6 is cleaved by regulated intramembrane proteolysis. The resulting amino-terminal fragment translocates to the nucleus and activates transcription by binding to ER stress-response elements present in the promoter regions of ER stress-inducible genes including those encoding ER chaperones and components of ER-associated degradation. The mammalian ATF6 family consists of two closely related homologs, ATF6α and ATF6β. ATF6α but not ATF6β plays a pivotal role in transcriptional control. The antibody was produced from hybridoma cultured in serum-free medium and purified under mild conditions by propriety chromatography processes.
Specifications:
Immunogen: Recombinant ATF6a (His-tagged amino-terminal fragment of ATF6a)
Isotype: Mouse IgG2a k
Form: Purified monoclonal antibody (IgG) 1mg/ml in PBS, 50% glycerol, filter-sterilized
Specificity: Specific to human ATF6a, no cross reactivity with mouse ATF6a
Storage: Shipped at 4℃ and stored at -20℃. (Avoid using freezer below -25℃)
Applications: (Detailed Protocol is given below)
Western blotting (1/500-1/1,000)
Immunoprecipitation (1/100-1/500)
Immunofluorescence staining (1/100-1/500)
Data Link:
UniProtKB/Swiss-Prot P18850 (ATF6A_HUMAN)
References: This antibody was used in Ref. 4 and described in Ref. 5.
Hai T et al “Transcription factor ATF cDNA clones: an extensive family of leucine zipper proteins able to selectively form DNA-binding heterodimers” Genes Dev 3: 2083-2090 (1989) PMID 2516827
Haze K et al “Mammalian transcription factor ATF6 is synthesized as a transmembrane protein and activated by proteolysis in response to endoplasmic reticulum stress” Mol Biol Cell 10: 3787-3799 (1999) PMID: 10564271
Yamamoto K et al “Transcriptional induction of mammalian ER quality control proteins is mediated by single or combined action of ATF6 and XBP1” Dev Cell 13: 365-376 (2007) PMID: 17765680
Maiuolo J et al “Selective activation of the transcription factor ATF6 mediates endoplasmic reticulum proliferation triggered by a membrane protein” PNAS 108: 7832-7 (2011) PMID: 21521793
Mori K “Divest yourself of a preconceived idea: transcription factor ATF6 is not a soluble protein!” Mol Biol Cell 21: 11435-8 (2010) PMID: 20219975
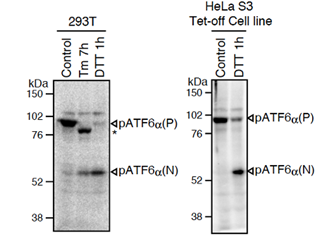
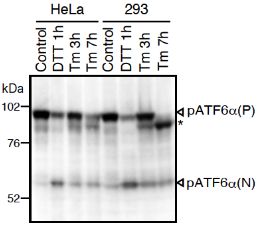
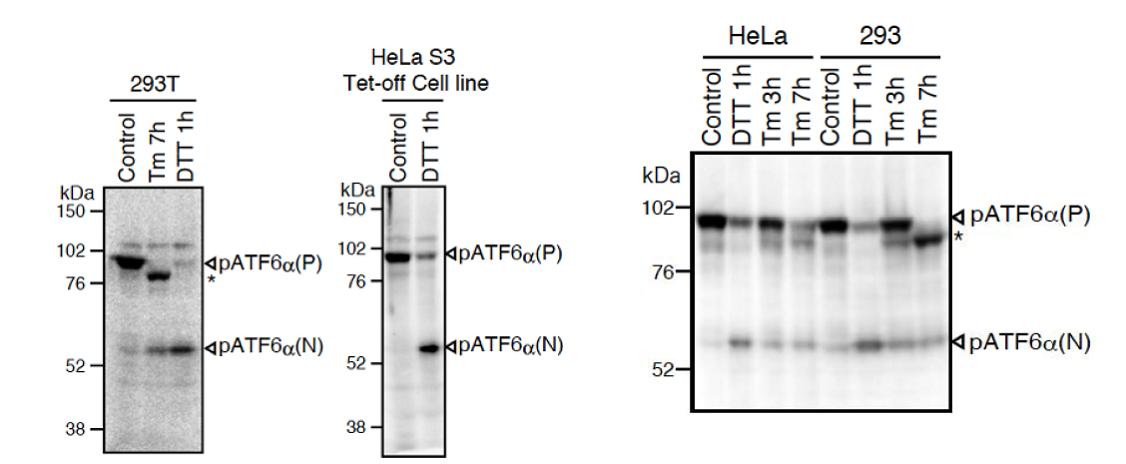
Fig.1 Western blot analysis of human cell extracts using this antibody: Conversion of pATF6a(P) to pATF6a(N) in DTT- or tunicamycin-treated cells. Tm: 2 mg/ml tunicamycin (inhibitor of N-glycosylation). DTT: 1mM dithiothreitol (reducing reagent). The asterisk denotes an unglycosylated form of pATF6a (P). ATF6a is constitutively expressed as pATF6α(P) (~90-kDa protein), and converted to pATF6a(N) (>50-kDa protein) in ER-stressed cells.
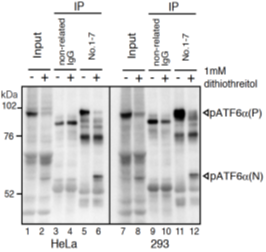
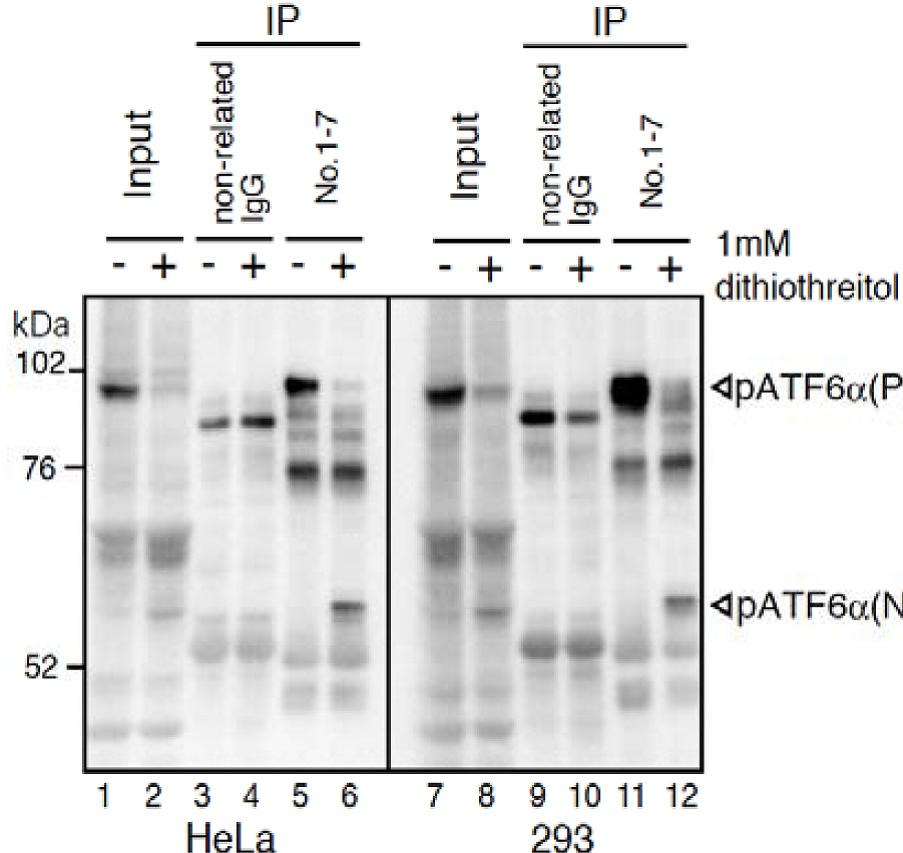
Fig.2 IP-Western blot analysis of human cell extracts using this antibody. ATF6a was detected by Western blot (Input; lanes 1, 2, 7, and 8) using this antibody (No.1-7). After immunoprecipitation (IP) with non-related IgG (IP; lanes 3, 4, 9, and 10) or this antibody (No. 1-7) (IP; lanes 5, 6, 11, and 12), samples were subjected to SDS-PAGE and analyzed by Western blot using this antibody (# 1-7) and anti-mouse IgG antibody (light chain specific). Detection of pATF6a(P) and pATF6a(N) is better in IP-Western blotting than Western blotting.
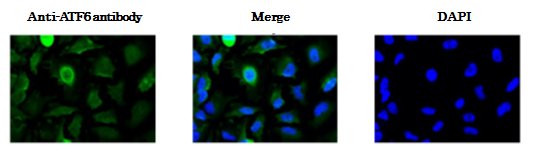
Fig.3 Immunofluorescence staining of ATF6 in HeLa cells by using anti-ATF6 antibody. The cells were fixed with paraformaldehyde. The antibody was used at 1/100 dilution. As the second antibody, AF488 conjugated goat anti-mouse IgG antibody was used at 1/1,000 dilution. In unstressed cell ATF6 is localized in endoplasmic reticulum and upon receiving stress signal it is proteolytically processed and translocated into nucleus where it functions as transcriptional activator.
Protocol for ATF6a analysis using anti-human ATF6a monoclonal antibody:
Both endogenous precursor ATF6a, pATF6a(P), and its cleaved product, pATF6a(N), can be detected in human cells such as HEK293T, HEK293 and HeLa cells by western blot analysis using anti-human ATF6a monoclonal antibody clone 1-7 (Fig. 1), according to the procedures described below. Clarity of the results may depend on cell types and culture conditions. If clear results could not be obtained by western blot analysis as seen with HeLa and HEK293 cells (Fig.1 right panel), it is worth while trying immunoprecipitation followed by western blot analysis according to the procedures described below.
Western blotting
SDS-sample buffer: 50 mM Tris/HCl, pH6.8, containing 2% SDS, (100 mM DTT), 10% glycerol and BPB
PBST: PBS containing 0.1% Tween 20 Blocking buffer: PBS containing 0.1% Tween 20 and 5% skim milk
Sample Preparation (for HeLa or HEK293 cells cultured in 6cm dish)
Wash cells with ice-cold PBS.
Scrape cells in 500 ml of ice-cold PBS (+ protease inhibitor cocktail and 10 mM MG132) 2 times and collect cells by centrifugation at 5,000 rpm for 2 min.
Lyse cells directly in 100 ml of SDS-sample buffer without reducing reagent (+ protease inhibitor cocktail and 10 mM MG132).
Vortex mix vigorously.
Boil the lysate for 5 min and vortex well.
If the lysate is still viscous, boil again and vortex mix vigorously.
Centrifuge at 14,000 rpm for 2 min.
Determine protein concentration using BCA protein assay kit.
SDS-PAGE and incubation with antibody
Add one-tenth volume of 1 M DTT and boil for 5 min.
Subject 50 mg of the lysate to 8% SDS-PAGE.
Transfer to nitrocellulose membrane (such as Hybond-ECL, GE Healthcare).
Incubate the membrane in Blocking buffer overnight at 4˚C.
Incubate the membrane with primary antibody diluted in Blocking buffer (1:500-1:1000) for 1 h at room temperature or overnight at 4˚C. Wash the membrane 3 times each for 5 min with PBST.
Incubate the membrane with HRP-conjugated secondary antibody for 1 h at room temperature. We recommend “ECL anti-mouse IgG, Horseradish Peroxidase linked F(ab’)2 fragment” (GE Healthcare NA9310V-1ML) or “Peroxidase-conjugated AffniPure Goat Anti-Mouse IgG, Light Chain Specific” (Jackson ImmunoReseach 115-035-174).
Wash the membrane 3 times each for 5 min with PBST.
Detect signals using an appropriate luminescent reagent.
Immunoprecipitation
(For HeLa or HEK293 cells cultured in 6 cm dish)
Lysis buffer: 50 mM Tris-HCl pH7.5, containing 150 mM NaCl, 1% NP-40, protease inhibitor cocktail
and 10 mM MG132)
Wash cells with ice-cold PBS, suspend them in 400 ml of Lysis buffer and stand on ice for 10 min.
Clear the lysate by centrifugation at 14,000rpm for 10 min at 4˚C and transfer 300 ml of the supernatant to a new tube.
Add anti-ATF6a antibody (1-3 mg) into the supernatant and incubate with gentle rotation for 2 h〜overnight at 4˚C.
Add 30 ml of the 50% slurry of ProteinG-Sepharose suspended in Lysis buffer into the tube and incubate with gentle rotation for 1 h at 4˚C.
Wash the Sepharose beads 2 times with Lysis buffer.
Wash the Sepharose beads with PBS.
Resuspend the Sepharose beads in 30 ml of SDS-sample buffer containing 100 mM DTT. Use 10 ml aliquot of the sample for Western blot analysis.
*Critical points
In IP-western blot analysis, use a light chain specific anti-mouse IgG antibody as a secondary
antibody. “Peroxidase-conjugated AffniPure Goat Anti-Mouse IgG, Light Chain Specific”
(Jackson ImmunoReseach 115-035-174) is recommended.


Reviews
There are no reviews yet.