Description
E.coli RuvA protein binds specifically to the Holliday structure which is the intermediate of recombination at the late stage of homologous recombination and recombination repair, and forms a complex with RuvB motor protein, allowing the migration of Holliday junction using ATP hydrolysis energy and expands the heteroduplex region. In solution, it forms a tetramer and binds to the cross-like DNA of the Holliday junction from below and above, holding it in between (1, 2). Using this antiserum in Western blot, the band of 22kD corresponding to RuvA was obtained from the extract of E. coli cells (Fig.1).
Applications
1) ELISA
2) Western blot x 3,000 dilution (Fig.1)
Other applications have not been tested.
Specification
Immunogen: Purified full-size recombinant RuvA protein (Ref. 2)
Form: Antiserum with 0.05% sodium azide
Storage: 4°C for short storage of 6 months. For long term storage, store at -80°C
DataLink: UniProtKB/Swiss-Prot P0A809 (RUVA_ECOLI)
References
- Shinagawa H and Iwasaki H (1996) “Processing the holliday junction in homologous recombination” Trends Biochem Sci 21:107-111PMID: 8882584
- Iwasaki H et al (1992) “Escherichia coli RuvA and RuvB proteins specifically interact with Holliday junctions and promote branch migration” Genes Dev 6:2214-2220 PMID: 1427081
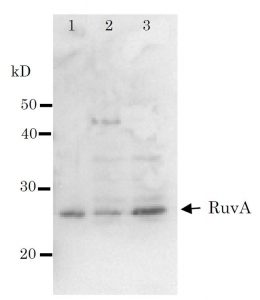
Fig. 1. Detection of RuvA (22kD) protein by Western blot using this antibody.
Lane 1: RuvA protein 0.8ng
Lane 2: E. coli AB1157 crude extract
Lane 3: E. coli AB1157 lexA mutant crude extract
Expression of RuvA is enhanced by lexA mutation.


Reviews
There are no reviews yet.