Description
The products of umuD, umuC, and recA genes (SOS genes) are required for mutagenesis induced by radiation or chemical agents. Transcription of these SOS genes is repressed by a repressor, LexA protein in uninduced cells (Ref.2). Exposure of cells to DNA-damaging agents activates RecA protein to promote proteolytic cleavage of LexA protein. Inactivation of LexA protein by the cleavage consequently derepresses the SOS genes, umuD, C and recA. UmuD protein is then auto-cleaved, which is promoted by RecA protein ssDNA in an ATP-dependent manner (Ref.1). The processed UmuD protein is the active form for mutagenesis and the UmuD-UmuC complex functions as an error-prone translesion DNA polymerase (Ref.3). The molecular weight of the intact UmuD is 17kD and the proteolytically processed active form is 14kD (Ref.1 & Fig.1).
Application
Western blot x 3,000 dilution (Fig.1)
Specification
Immunogen: Purified recombinant LacZ’-UmuD fusion protein
Form: Antiserum with 0.05% sodium azide
Storage: Shipped at 4°C and store at -20°C or long term storage, -70°C
Data Link
Swiss-Prot P0AG11
References
This antibody was used in Ref.1.
- Shinagawa H et al (1988) “RecA protein-dependent cleavage of UmuD protein and SOS mutagenesis.” Proc Natl Acad Sci USA 85: 1806-1810 PMID: 3126496
- Kitagawa Y et al (1985) “Structural analysis of the umu operon required for inducible mutagenesis in Escherichia coli.” Proc Natl Acad Sci USA 82: 4336-4340 PMID: 2989817
- Friedberg EC et al DNA Repair and Mutagenesis 2nd ed., ASM Press
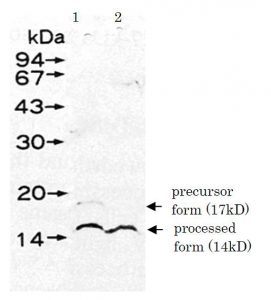
Fig. 1. Detection of UmuD protein in the extract of E. coli DE274 (lexA51, recA730) by Western blot using this antibody.
Lane 1: without mitomycin C treatment
Lane 2: treated with mitomycin C


Reviews
There are no reviews yet.