Description
EDD / UBR5 (2,799 aa, 309 kDa) is an E3 ubiquitin-protein ligase which is a component of the N-end rule pathway. It recognizes and binds to proteins bearing specific N-terminal residues that are destabilizing according to the N-end rule, leading to their ubiquitination and subsequent degradation. EDD/UBR5 is involved in maturation and/or transcriptional regulation of mRNA by activating CDK9 by polyubiquitination and may play a role in the control of the cell cycle progression. The ligase may have a tumor suppressor function. It regulates DNA topoisomerase II binding protein (TopBP1) in the DNA damage response. It plays an essential role in extra embryonic development. Ubiquitinates acetylated PCK1. Also acts as a regulator of DNA damage response by acting as a suppressor of RNF168, an E3 ubiquitin-protein ligase that promotes accumulation of ‘Lys-63’-linked histone H2A and H2AX at DNA damage sites, thereby acting as a guard against excessive spreading of ubiquitinated chromatin at damaged chromosomes.
Applications
1. Western blotting (1/1,000 dilution, Fig. 1)
Specification
Immunogen: Synthetic peptide of human EDD protein corresponding to 394-408 amino acids, C-KWSESEPYRNAQNPS, conjugated with KLH
Reactivity: Human (HeLa, HEK293T, MCF7) and mouse (NIH3T3). Note that expression level of EDD varies greatly, depending on tissues and cell types (see Fig.1). Not tested in other species. Form: 1 mg/ml in 1x PBS, 50% glycerol. Filter-sterilized. Azide and carrier free.
Product: Affinity-purified from rabbit antiserum with immunogen peptide conjugated with agarose beads.
Storage: Shipped at 4°C or at -20°C. Upon arrival, spin-down and store at -20°C.
Data Link: uniprot/O95071 UBR5 human
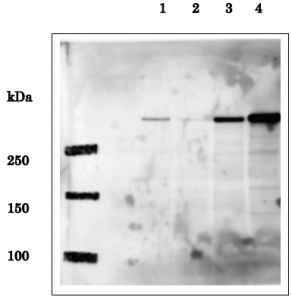
Fig.1 Identification of EDD in whole human cell extracts by western blot using anti-EDD antibody.
Lane 1: HeLa cells transfected with control siRNA
Lane 2: HeLa cells transfected with EDD-specific siRNA Lane 3: HEK293T cells transfected with pFlag-CMV-5b empty vector
Lane 4: HEK293T cells transfected with pCMV-Tag2b-EDD expression vector.
Predicted molecular mass of EDD is 309 kDa and the corresponding band in Lane 1 is much reduced in Lane 2 by introduction of the EDD-specific siRNA.
Note that expression level of EDD in HEK293T cells is much higher than in HeLa cells. The data are by courtesy of Prof. M. Fujita of Kyushu University
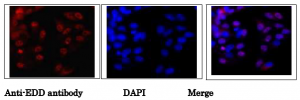
Fig.2. Immunofluorescence staining of EDD protein in MCF7 cells.
MCF7 cells were fixed in 4% paraformaldehyde overnight and permeabilized in 0.25% TritonX 100 in PBS for 10 min. Anti-EDD antibody was used at 1/1,000 dilution. As second antibody, goat anti-rabbit IgG conjugated with Alex488 was used at 1/1,000 dilution. DNA was stained with DAPI. Note that not all nuclei were stained, indicating cell cycle dependency of EDD protein existence.


Reviews
There are no reviews yet.