Description
p53 mutants are found in more than half of human cancers and are considered as the most important human cancer related gene. p53 is detected at 53kD position by electrophoresis and is composed of 393 amino acids. In the unstressed normal cells, the p53 level is low and it is inactive. However, with stress, especially with DNA damage, it is activated to promote arrest of cell cycle and repair of DNA damage, or induction of apoptosis. The functions of p53 are regulated by phosphorylation of serine and threonine, and acetylation of lysine at various sites in the molecule (1). p53 at Ser20 is phosphorylated by Chk1 and Chk2 kinases and this enhances tetramerization, stability and activity of p53 (2, 3).
Applications
1) Western blot ~ 1ug/ml 2) ELISA
Specification
Antigen: synthetic peptide containing phosph-Ser20
Isotype: mouse IgG1 Kappa
Form: purified monoclonal antibody (IgG) 1mg/ml in PBS, 50% glycerol, filter-sterilized
Reaction: human p53 phosphorylated at Ser20
Storage: -20 °C, for long term storage -70°C
Data Link: UniProtKB/Swiss-Prot P04637 (P53_HUMAN)
References
1.Bode AM & Dong Z “Post-translational modification of p53 in tumorigenesis”Nature Rev Cancer 4: 793 -805 (2004) PMID: 15510160
2.Shieh SY et al “DNA damage-inducible phosphorylation of p53 at N-terminal sites including a novel site, Ser20, requires tetramerization”EMBO J 18: 1815-1823 (1999) PMID: 10202145
3.Hirao A “DNA damage-induced activation of p53 by the checkpoint kinase Chk2” Science 287: 1824-1827 (2000) PMID: 10710310
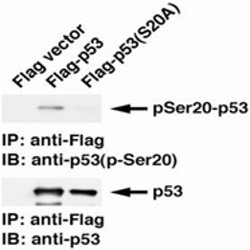
Fig.1 Identification of p53 phospho-Ser20 with monoclonal antibody 17B6 by Western blot.
The lysates of H1299 cells (p53 non-producing) transfected either with Flag vector (left lane), Flag-p53 (wild-type) expressing plasmid (middle lane) or Flag-p53 mutant (Ser20Ala) expressing plasmid (right lane) were immunoprecipitated with anti-Flag antibody and then analyzed by Westernbotting with anti-p53 p-S20 monoclonal antibody, 17B6, (upper panel) or with pantropic p-53 antibody (lower panel).


Reviews
There are no reviews yet.