Description
Background:
E. coli RecA protein is a very important enzyme for homologous recombination and recombinational repair. Its synthesis is induced by SOS response caused by DNA damage. RecA protein has multiple functions such as single stranded DNA dependent ATPase activity, DNA annealing activity, formation of D-loop and Holliday structure in homologous recombination reaction, and coprotease activities that promote self-cleavages of LexA repressor, lambda phage repressor and UmuD protein. RecA protein binds to single and double stranded DNA for nucleofilament formation. It carries out a central role in homologous recombination. Its homologs in eukaryotes are Rad51 protein and Dmcl protein (2).
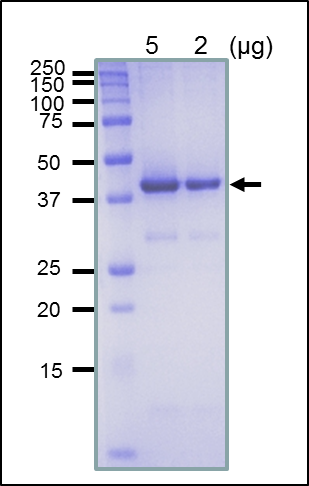
The product is over-expressed as a recombinant protein and highly purified by several steps of chromatography. A single band is observed by SDS-PAGE at 38 kD (Figure).
Specifications:
Storage: Shipped at 4°C or -20℃ and store at -20℃. (-80°C for long-term storage).
Form: 1.0 mg/ml in 20 mM Tris-HCl (pH 8.0), 1 mM EDTA, 150 mM KCl,1 mM DTT, 50% glycerol
Purity: Over 90% by SDS-PAGE (CBB staining)
Applications
1) Studies on homologous recombination mechanism and SOS response.
2) Useful in the screening using probe from library by promotion of DNA hybridization (1).
3) Facilitate DNA observation by electron microscope due to nucleofilament formation with DNA.
Data Link UniProtKB/Swiss-Prot P0A7G6 (RECA_ECOL)
References:
This protein was used in the publications below:
1. Horii T et al.Regulation of SOS functions: purification of E. col LexA protein and determination of its specific site cleaved by the RecA protein Cell. 27:515-22. (1981) PMID: 6101204
2. Oura S et al Biomolecular recognition ability of RecA proteins for DNA single-walled carbon nanotubes. Colloids Surf B Biointerfaces. 2015 Feb 1;126:496-501.)


Reviews
There are no reviews yet.