Description
Description
HIV-1 protease is the aspartyl protease that mediates proteolytic cleavages of Gag and Gag-Pol polyproteins during or shortly after the release of the virion from the plasma membrane. Cleavages take place as an ordered, step-wise cascade to yield mature proteins. This process is called maturation. HIV-1 protease displays maximal activity during the budding process just prior to particle release from the cell. It also cleaves Nef and Vif, probably concomitantly with viral structural proteins on maturation of virus particles. Hydrolyzes of the host EIF4GI and PABP1 turns off the capped cellular mRNA translation process, resulting in the inhibition of cellular protein synthesis and ensures maximal viral gene expression and evasion of host immune response.
Full-size functional recombinant HIV-1 protease purified from E. coli.
Usage
- Functional studies such as screening for inhibitors of HIV-1 protease for AIDS drug discovery
- Antigen for anti-HIV 1 antibody for Western blotting and ELISA
- Control as HIV 1 protease for SDS-PAGE
Other applications have not been tested.
Specification
Stock solution: 0.2 mg /ml in 20 mM Tris, 20 mM MES, 0.2 M NaCl, 1 mM EDTA, 1 mM DTT, 10% Glycerol, pH 6.5
Reaction buffer: 20 mM Tris-HCl (pH 6.8), 1 mM EDTA, 1 mM DTT, 0.1% Triton X-100, 10% Glycerol
Shipped with dry-ice and stored at -80°C.
When necessary, freeze (with liquid or ethanol in dry-ice) and thaw (in water at room temperature with shaking) should be done rapidly.
Data Link
UniProt P03367 (gag-pol), UniProt Q9YQ30 (HIV-1 Protease)
Reference
The HIV-1 strain and the recombinant protease has been described in the following references.
- Adachi A et al “Production of acquired immunodeficiency syndrome-associated retrovirus in human and nonhuman cells transfected with an infectious molecular clone” J Virol 59: 284 -291(1986) PMID: 3016298
- Saitoh A et al “Overproduction of human immunodeficiency virus type I reverse transcriptase in Escherichia coli and purification of the enzyme” Microbiol Immunol 34:509-521 (1990) PMID: 16991
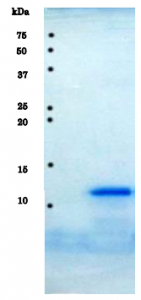
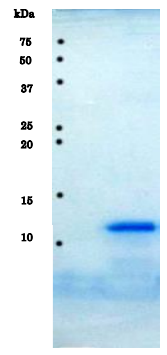
Fig.1 SDS-PAGE analysis of purified HIV-1 protease.
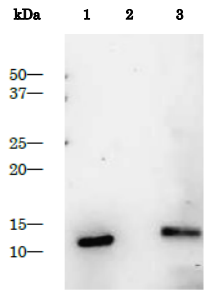
Fig. 2 Purified HIV-1 protease as control antigen for Western blot analysis with anti-HIV-1 protease antibody.
Lane 1: Purified HIV-1 protease (1 ng), Lane 2: Extract of MT4 cells, Lane 3: Extract of MT4 cells infected with HIV-1 (LAI strain). The anti-HIV-1 protease antibody (Catalog # 65-018) was used at 1/2,000 dilution.
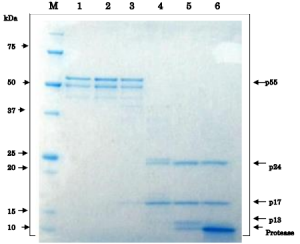
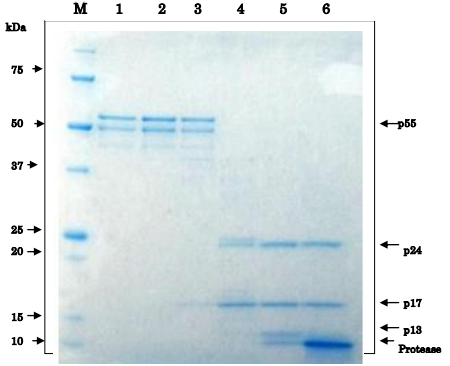
Fig.3 Proteolytic processing of HIV-1 Gag p55 proprotein by HIV-1 protease in vitro
As the substrate, recombinant Gag p55 (1 μg, Catalog # 05-009) was used in 20 μl reaction volume. The reaction was carried by incubating at 37°C for 3 hr and stopped by adding SDS-PAGE sample buffer. Lane 1: no protease, Lane 2: 0.16 pg, Lane 3; 1.6 pg, Lane 4: 16 pg, Lane 5: 0.16 μg, Lane 6: 1.6 μg protease. Note that two degradation bands are observed in the preparation of p55 substrate. In lane 4, p25 band is visible and in lane 5, p13 band is visible.
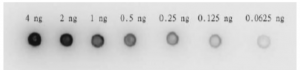 Fig.4a
Fig.4a
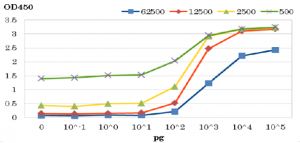 Fig.4b
Fig.4b
Fig.4a. Dot blot of HIV-1 protease by using anti-HIV-1 protease antibody. Anti-HIV-1 protease antibody (Catalog # 65-018) was used at 1/2,000 dilution. As second antibody, goat anti-rabbit IgG antibody conjugated with HRP was used at 1/5,000 dilution.
Fig.4b ELISA of HIV-1 protease with anti-HIV-1 protease antibody (Catalog # 65-018). The antibody was used at dilutions indicated above. Purified Protease was spotted in wells.


Reviews
There are no reviews yet.