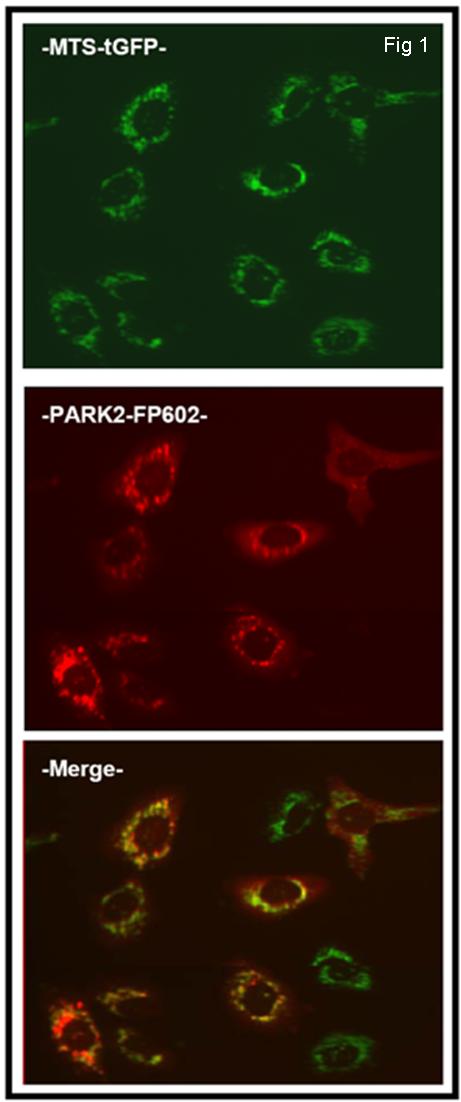
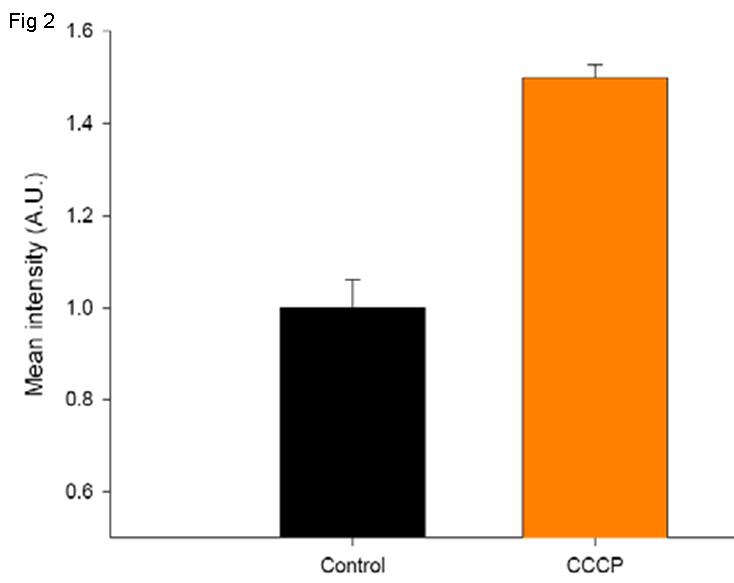
Description
Parkinson’s disease (PD) is a neurodegenerative disorder caused by the death of dopamine producing cells in the substantia nigra in the brain. The main sympton is trembling in the hands, legs or face. Most cases of PD are sporadic, but 10% are inherited either in an autosomal dominant or recessive manner. Parkin is one of the proteins implicated in one form of familial PD known as autosomal recessive juvenile Parkinson’s disease (AR-JP). Parkin is a protein that forms part of E3 ubiquitin ligase complex that mediates the targeting of proteins for degradation. Parkin is recruited to damaged mitochondria during the early stages of mitophagy induced by CCCP.Innoprot has developed a PARK2-FP602+MTS-tGFP U2OS Stable Cell line, which has been obtained by a double transfection of an expression vector that encodes turbo green fluorescent protein (tGFP) fused to mitochondrial targeting sequence (MTS) derived from the subunit VIII of human cytochrome C oxidase and a vector containing PARK2 protein fused to FP602 red fluorescent protein.Red fluorescent Parkin is recruited to damaged green fliorescent mitochondria during the early stages of mitophagy induced by CCCP. This cell line has been designed to ensure the colocalization of PARK2-FP602 and Mitochondria once CCCP has been added.To analyze PARK2 mitochondrial localization, a Region Of Interest (ROI) of PARK2 is delimited and the intensity of mitochondria signal (GFP) is quantified in this ROI. So, when PARK2 is recruited in the mitochondria, the intensity of the GFP is increased.Applications:This model can be used in drug discovery for compounds that inhibit mitochondrial loss of membrane potential through parkin recruitment. This model is a usefull tool to test possible drugs against Parkinson’s disease.This model allows to analyse in the space and time the compund effect in a multiparametric manner.