Description
Background:
Streptolysin O (SLO) is a membrane-damaging extracellular toxin produced by hemolytic streptococci. The membrane-damaging activity is measured by hemolysis of red-blood cells. SLO is easily inactivated in the presence of oxygen but can be reactivated by thiol compounds, so it is also called thiol-activated cytolysin . SLO is produced not only by Group A hemolytic streptococci but also by Group C and Group G strains. The amino acid sequences are highly conserved among them and their homology is over 98%.
Specifications:
Product: Recombinant streptolysin O. Functional in membrane pore formation to introduce molecules into living animal cells. The product was highly purified from E.coli over-expressing SLO of Group C hemolytic streptococci (His6-tagged to the signal peptide removed N-terminal of SLO). The specific activity is as high as 1,200,000- 2,000,000 hemolytic units (HU) /mg (depending on Lot).
Measurement of the activity: Definition of 1HU is activation of 50% hemolysis by incubating 3% sheep red blood cells at 37℃ for 30 min.
Purity: Over 98% by SDS-PAGE (see Fig.1)
Form: 1 mg/ml in PBS (-), 1 mM DTT, 50% glycerol, sterilized by filtration. No additive nor carrier protein. Inactivated SLO can be reactivated by thiol reagents such as 20 mM cysteine or 10 mM DTT (Palmer M “The family of thiol-activated, cholesterol-binding cytolysins.” Toxicon 39: 1681-1689 (2001) PMID: 11595631 )
Storage: Shipp at 4℃ or -20℃, and store at -20℃ (For longer period, years, store at -80℃)
Applications
- Antigen for the measurement of anti-streptolysin O antibody (ASO) (diagnostic reagent), ELISA
- Western blotting, Dot blotting,
- Immuno-chromatography
- SDS-PAGE
- Reagent for membrane pore formation to introduce small-to-macromolecules into living cells (For protocol refer to or other references; Walev I et al “Delivery of proteins into living cells by reversible membrane permeabilization with streptolysin-O.” PNAS 98: 3185-3190 (2001) PMID: 11248053)
Data Link: UniProtKB Q54114 ((TACY_STREQ)
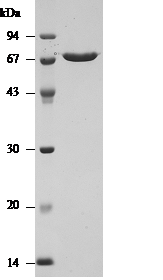
Fig. 1 Purified SLO analysed by SDS-PAGE.
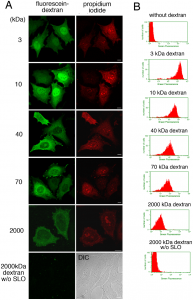
Fig.2 Introduction of fluorescein dextran of different molecular weights into resealed cells. A. HeLa cells were incubated with or without (2000 kDa dextran w/o SLO) 0.13 µg/ml SLO on ice for 5 min. After wash with PBS three times, the cells were further with transport buffer containing propidium iodide at 32°C for 5 min. Semi-intact HeLa cells were incubated with 1.5 mg/ml L5178Y cytosol, an ATP regenerating system, GTP, glucose, and 100 µg/ml fluorescein-dextran of 3, 10, 40, 70, or 2000 kDa at 32°C for 15 min, and then were resealed by treatment with 1 mM CaCl2 at 32°C for 5 min. After incubation with DMEM supplemented with FCS for 30 min, the cells were observed by confocal microscopy. Since the cells without SLO treatment did not contain the fluorescence of propidium iodide, differential interference contrast (DIC) image was shown. Bar = 10 µm. B. HeLa cells were treated as described in A, were trypsinized, and were subjected to flowcytometry. The histograms of fluorescein fluorescence of dextran with different molecular weight in PI-positive cells were shown.
Data from Kano F. et al. PLoS One. 2012;7(8)
http://dx.doi.org/10.1371/journal.pone.0044127.g003
References:This product has been used in the following publications.
- Maeda, Y. et al. GPHR is a novel anion channel critical for acidification and functions of the Golgi apparatus. Nat. Cell Biol. 10: 1135-45 (2008) PMID: 18794847 Permeabilization of cells.
- Furukawa K. et al. Reduction-triggered fluorescent amplification probe for the detection ofendogenous RNAs in living human cells. Bioconjug Chem.2009 May 20;20(5):1026-36. PMID: 19374406 Introduction of probes for RNA into permeabilized human HL cells
- Thiery J. et al. Perforin activates clathrin- and dynamin-dependent endocytosis, which is required for plasma membrane repair and delivery of granzyme B for granzyme-mediated apoptosis. Blood 2010 115:1582-1593. PMID: 20038786 Promotion of endocytosis
- Kano F. et al. Hydrogen peroxide depletes phosphatidylinositol-3-phosphate from endosomes in a p38 MAPK-dependent manner and perturbs endocytosis. Biochim Biophys Acta.2011 May;1813(5):784-801. PMID: 21277337. Permeabilization of HeLa cells.
- Potez S. et al. Tailored protection against plasmalemmal injury by annexins with different Ca2+ sensitivities. J Biol Chem.2011 May 20;286(20):17982-91. PMID:
- 21454475 Permeabilization of HEK cells.
- Kano F. et al. A resealed-cell system for analyzing pathogenic intracellular events: perturbation of endocytic pathways under diabetic conditions. PLoS One.2012;7(8):e44127. PMID: 22952896 Introduction of molecules into HeLa cells.
- Imai A. et al. MADD/DENN/Rab3GEP functions as a guanine nucleotide exchange factor for Rab27 during granule exocytosis of rat parotid acinar cells. Arch Biochem Biophys.2013 Aug 1;536(1):31-7. PMID: 23702376. Introduction of antibody into cell
- Gao N and Lehrman MA. Mannose-6-Phosphate: A Regulator of LLO Destruction. : Inka Brockhausen (ed.), Glycosyltransferases: Methods and Protocols, 2013, Springer, Methods in Molecular Biology, vol. 1022, DOI 10.1007/978- Inka Brockhausen (ed.), Glycosyltransferases: 1-62703-465-4_20. Link:.springer.com/protocol/10.1007 Introduction of mannose-6-phosphate into living cells. (The authors specifically recommend BioAcdemia streptolysin O for cell permeabilization)
- Matsuto M et al. Reconstitution of the targeting of Rab6A to the Golgi apparatus in semi-intact HeLa cells: A role of BICD2 in stabilizing Rab6A on Golgi membranes and a concerted role of Rab6A/BICD2 interactions in Golgi-to-ER retrograde transport. Biochim Biophys Acta.2015 Oct;1853(10 Pt A):2592-609. PMID: 25962623 Introduction of protein (Rab6A) into permeabilized Hela cells.
- Yasuga H. et al. Logic gate operation by DNA translocation through biological nanopores. PLoS One.2016 Feb 18;11(2):e0149667. PMID: 26890568 Nanopore formation in bilayer lipid menmbrae.

