Description
Thermus aquaticus RecA protein is a thermostable enzyme which plays important roles in homologous recombination and DNA repair. This protein has activities of single-stranded DNA dependent ATPase, DNA annealing, and exchanging of strands between two recombining DNA double helices, similar to E. coli RecA protein, but the optimal temperature is between 65~75°C (1). Taq RecA was expressed in E. coli in large quantities and the protein was then purified. MW is 36.5kD.
Applications
- Useful for studying homologous recombination
- Increase the specificity and yield of multiplex PCR (of cDNA or genomic DNA) by promoting homologous annealing of primers to target DNA (2)
- Visualization of DNA with electron microscopy due to nucleofilament formation.
Specification
Form: 1 mg/ml in 50mM Tris-HCl (pH 8.0), 200mM NaCl, 1mM EDTA, 50% glycerol
Store: -20°C
Activity: The activity of single-stranded DNA-dependent ATPase was confirmed.
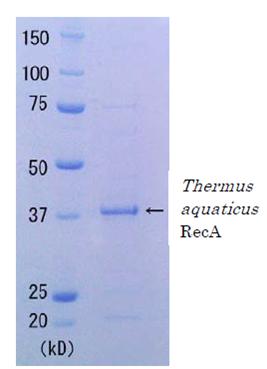
Quality: Single-strand dependent ATPase activity. Greater than 90% of protein determined by SDS-PAGE (Fig. 1 CBB staining). The absence of endonucleases and exonucleases was confirmed.
Data Link: UniProtKB/Swiss-Prot P48296 (RECA_THEAQ) P48296
References:
- Angov E & Camerini-Otero RD (1994) “The recA gene from
the thermophile Thermus aquaticus YT-1: cloning, expression,
and characterization.” J. Bacteriol. 176: 1405-1412 PMID: 8113181
- Shigemori Y et al (2005) “Multiplex PCR: use of heat-stable
Thermus thermophilus RecA protein to minimize non-specific
PCR products.” Nucleic Acids Research 33: e126 PMID: 16087733
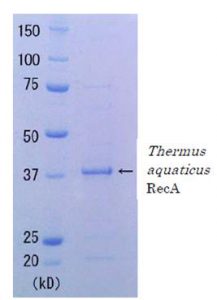
Fig. 1 SDS-PAGE of Thermus aquaticus RecA protein
Related products:


Reviews
There are no reviews yet.