Description
Glutathione S transferase (GST) from Schistosoma japonicum is commonly used to create fusion proteins. GST-tag has the size of 220 amino acids (roughly 26kDa) and is fused to the N-terminus of a protein. GST fusion proteins can be produced in Escherichia coli, as recombinant proteins and are used to purify and detect proteins of interest. The GST part binds its substrate, glutathione. GST-fusions protein can be easily purified from cell extracts by affinity chromatography with glutathione resin.
Applications
- Western blot dilution: 1/2,000~1/10,000
- Immunoprecipitation assay dependent
- ELISA
Other applications have not been tested
Specification
Immunogen: Recombinant full-size GST (1-212 aa)
Form: Antiserum with 0.05% sodium azide
Specificity Specific to GST and GST-tagged proteins
Storage: -20°C or for long term storage, -70°C
Data Link
NCBI Protein Data AAA57089
References
- Smith DB & Johnson KS (1988) “Single-step purification of polypeptides expressed in Escherichia coli as fusions of glutathione-S-transferase.” Gene 67:31-40 PMID: 3047011
- Kaelin WG Jr et al (1991) “Identification of cellular proteins that can interact specifically with the T/E1A-binding region of the retinoblastoma gene product.” Cell 64:521-532 PMID: 1825028
- Molecular Cloning: A laboratory Manual (eds. Sambrook, J., Russell, D.W. Cold Spring Harbor Laboratory Press, Cold Spring Harbor, New York, USA, 2001) pp.15.36-15.39, pp.18.48-18.59.
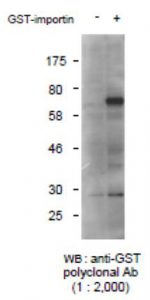
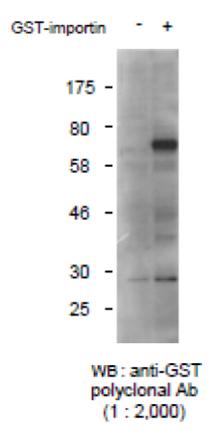
Fig.1 Detection of GST-tagged protein with this antibody by Western blotting.
-: Lysate of 293T cells transfected with an empty vector
: Lysate of 293T cells transfected with the plasmid carrying
the GST-tagged importin gene
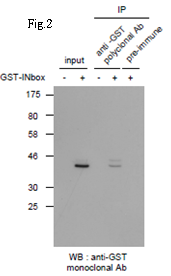
Fig.2 Immunoprecipitation of GST-tagged protein with this antibody followed by Western blot.
-: Lysate of 293T cells transfected with an empty vector
: Lysate of 293T cells transfected with the plasmid carrying
the GST-tagged INbox gene


Reviews
There are no reviews yet.