Description
Caspases are a family of cysteine proteases which play essential roles in apoptosis. Among them, Caspase 3 is a frequently activated death protease, catalyzing the specific cleavage of many key cellular proteins. Caspase 3 is synthesized as an inactive 32 kDa pro-enzyme which undergo proteolytic processing in response to apoptotic stimulation to produce the active form which consists of the p20/p17, and p12 subunits. Caspase 3 is the predominant caspase involved in the cleavage of Alzheimer amyloid precursor protein (APP), which is associated with neuronal death in Alzheimer‘s disease. An antibody (named ACP3) against activated caspase 3 was raised in rabbit. This antibody recognizes the active form of human caspase 3, p20/p17 subunit but does not recognize the proenzyme p32 (ref. 5).
Applications
Western blot (dilution: 1/3,000 – 1/1,000)
Immunocytochemistry (dilution: 1/1,000 – 1/500)
ELISA
These applications were confirmed in the laboratory of Prof. K, Yoshikawa of Osaka University (ref. 3).
Specification
Immunogen: Synthetic peptide corresponding to the caspase 3 cleavage site, 6 aa (CGIETD)
Specificity: Specific to the activated caspase 3 of human, mouse and rat. The antibody does not react with the proenzyme p32.
Form: Antiserum with 0.05% sodium azide
Storage: Shipped at 4°C and store at -20°C
Data Link:
UniProtKB/Swiss-Prot P42574 (CASP3_HUMAN)
References:
Thornberry NA and Lazebnik Y (1998) “Caspases: enemies within” Science 281: 1312-1316 PMID: 9721091
Uetsuki T et al (1999).”Activation of neuronal caspase-3 by intracellular accumulation of wild-type Alzheimer precursor protein” J Neurosci 19: 6955-6964 PMID: 10436052
Nishimura I et al (2002) “Cell death induced by a caspase-cleaved transmembrane fragment of the Alzheimer amyloid precursor protein” Cell Death Differ 9: 199-208 PMID: 11840170
Nishimura I et al. (2003) “Up regulation and antiapoptotic role of endogenous Alzheimer amyloid precursor protein in dorsal root ganglion neurons” Exp Cell Res 286: 241-251 PMID: 12749853
Kouroku Y et al (1998) “Detection of activated caspase-3 by a cleavage site-directed antiserum during naturally occurring DRG neurons apoptosis.” Biochem Biophys Res Comm 247: 780-784 PMID: 9647770
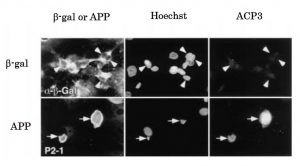
Fig.1 Immunocytochemistry for APP, chromosomal DNA, and activated caspase 3 subunits : Caspase 3 activation in neurons accumulating wild-type APP (ref.3). NT2 neurons (neurally differentiated human NT2 embryonic carcinoma cells) were infected with adenovirus vector expressing β-galactosidase (upper panel) or APP (lower panel), fixed 48 h later, and triply stained for the N-terminus of APP (with antibody P2-1) or β-gal (with antibody against β-gal), chromosomal DNA (Hoechst), and activated caspase 3 subunits (with antibody ACP3). Some neurons accumulating APP are strongly immunostained with ACP3 (arrows), whereas neurons accumulating β-gal are hardly labeled (arrowheads).
Related products:
# 74-104 anti-APP (C-terminus) antibody
# 74-106 anti-APP (N-terminus) antibody
# 74-108 anti-APP (C-terminus of the caspase3-cleaved APP) antibody
# 74-110 anti-APPΔ31 (specific to C-terminal APPΔ31) antibody


Reviews
There are no reviews yet.